LLAMA ANTIBODY RESEARCH
LLAMAS IN THE LAB
Camelids – Llamas, Alpacas, Guanacos, Vicuñas, and Camels are unique in that they have
specialized antibodies circulating in their blood. The 1989 discovery of a naturally small antibody in a camels blood has led to the development of new treatments to fight C. difficile,
hepatitis B, HIV, cancer and Alzheimer’s disease.
Current research being conducted by Dr. Jamshid Tanha’s antibody therapeutics group at the National Research Council Canada has revealed how antibodies derived from llamas may provide a new method for diagnosing and treating Alzheimer’s Disease.
Dr. Tanha has been involved in llama antibody therapeutics since 1997. Scientists with the NRC, in collaboration with the University of Calgary, have shown how camelid antibodies recognize disease toxin proteins. This research is an important step towards developing specific ways to to treat very common and serious diseases.
The NRC is based in Ottawa and has three integrated research and development divisions. The sectors – Emerging Technologies, Engineering, and Life Sciences, represent areas of economic
and scientific value for Canada. The Life Sciences sector develops human health technologies including vaccines, immunotherapeutics, and therapeutics that can cross the blood-brain barrier; research involving anti-bodies derived from llamas.
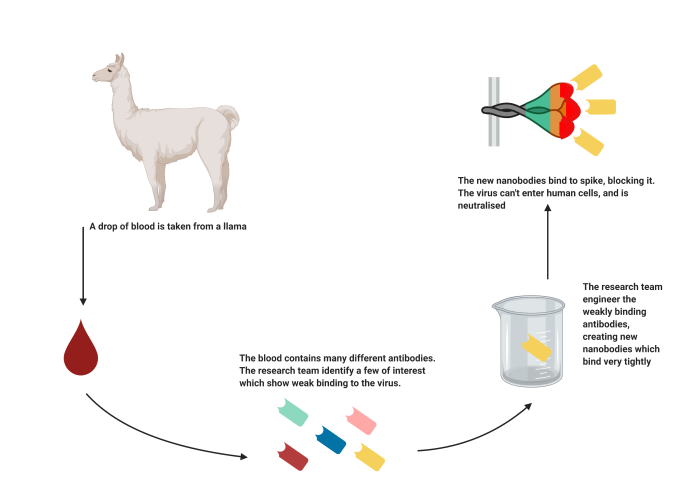
Llamas have normal antibodies like our own, as well as heavy chain antibodies from which single-domain antibodies are derived. These antibodies are ten times smaller than those found in humans and can be more readily engineered into drug therapies. Their small size enables them to reach places that our normal antibodies can’t. The single domain nature of these antibodies provide unique advantages which set them apart for research, diagnostic and therapeutic applications.
The discovery of this antibody came about by chance. In 1989, students at the Free University
of Brussels had identified what they thought were antibody fragments in a camel blood sample.
A professor at the university, Raymond Hamers, and his colleagues were intrigued, and demonstrated that the antibodies found in camels, ( and other members of the camelid family),
consisted of two heavy chains, instead of two heavy and two light chains that make up the antibodies produced by humans.
According to Mehdi Arbabi Ghahroudi of the NCR, these unique antibodies account for 70 percent of the antibodies in a camel’s blood and 30 to 40 percent in a llama’s. As llamas are smaller, easier to work with, and more readily available than camels, companies that supply
the blood samples prefer to work with them.
HOW DO ANTIBODIES WORK?
An antibody is a natural protein produced by white blood cells when mounting an immune response to a foreign substance (antigen) introduced into the body, such as bacteria or viruses. The antibody latches onto the antigen and either destroys it or makes it easier for white blood cells to eliminate the unwanted foreign substance. Vaccines help prepare the body, so it can
react faster and more effectively to a pathogen invasion.
ADVANTAGES OF LLAMA ANTIBODIES
The smaller size allows them to penetrate tissues, pass through the blood-brain barrier and bind epitopes that cannot be reached by conventional antibodies, making them excellent to use in immunohistochemistry.
Llama antibodies are capable of recognizing a variety of epitopes, (part of an antigen that the antibody recognizes and attaches to), located not only on the surface of a protein, but also in
the clefts.
They have a high tolerance to extreme pH and temperature. They are more durable, capable
of withstanding temperatures of almost 200 degrees Fahrenheit.
They are easy to clone, hence a cheaper alternative for large scale production.
ALZHEIMER’S DISEASE: LLAMA ANTIBODIES DETECT CEREBRAL LESIONS
A number of research teams from around the world have been working with the NRC todevelop effective therapies and diagnostics involved with llama-derived antibody proteins.
The development of these “nanobodies” have important applications in Alzheimer’s disease.
These nanobodies are capable of crossing the blood-brain barrier, (which generally protects the brain from microbial attacks but also prevents potential therapeutic molecules from reaching it), and once in the brain can specifically mark and show amyloid plaques and neurofibrillary tangles, the two types of lesions that characterize Alzheimer’s disease.
Amyloid beta peptide, naturally present in the brain, builds up over the years as a result of genetic and environmental factors until it forms amyloid plaques. This build-up is toxic for nerve cells and leads to a loss of neuronal structure and to what is known as neurofibrillary tangles, which in turn results in cell death.
“The blood-brain barrier rejects many things, including drugs”, says Dr. Tanha. “We’ve developed single-domain antibodies that are ‘transcytotic’, which means they can get across
the cells of the brain’s protective layer.”
Early detection of Alzheimer’s can lead to better treatment and better outcomes for peoplewith this brain disease. The scientists involved in this collaborative project are working on
MRI techniques to detect the lesions associated with Alzheimer’s disease. At present they are
using two-photon microscopy to detect signs of the disease in vivo in mice. Human trials using llama antibodies in Alzheimer’s patients are predicted to get underway in the next few years.
With greater interest and involvement from the pharmaceutical and biotechnology companies
llama-derived antibodies may soon become a standard therapy in testing, diagnosing, and treating diseases in humans as well as animals. Dow Agro Sciences has licensed llama antibody technology for animal health and food safety applications. Texas Biomedical Research Institute is developing tests using llama-derived antibodies to detect hemorrhagic fever viruses, including Ebola and Marburg.
EVOLUTIONARY ADVANTAGES OF THE CAMELID FAMILY ARE PROVING TO BE OUR NEW LINE OF DEFENCE – IN SO MANY WAYS
Llama antibodies can act as sensors to warn of dangerous microbes and chemicals in the event of a biological attack. Researchers at the U.S. Naval Research Laboratory in Washington were able to successfully engineer llama-derived antibodies that have the ability to detect cholera toxin, a smallpox virus surrogate, and ricin in the environment. Scientists at Southwest Foundation for Biomedical Research have developed tests using llama-derived antibody proteins to detect seven different types of botulinum neurotoxins. Botulinum neurotoxins are about 100 billion times more toxic than cyanide, and should there be a bioterror threat, the ability to detect these toxins in our water supply or food-chain is critically important.
CUSTOM ANTIBODIES
About a dozen Science and Technology companies are now offering single-domain antibody
services. Using bioengineering techniques, the antibody genes from llama and alpaca blood samples are cloned. These “libraries” provide researchers with naive and immunized camelid antibodies from which they can conduct their research and develop new techniques to safeguard human health, target certain diseases, and discover new cures.
Most of us know llamas as providers of luxurious fleece, or for their athletic abilities as a
pack-animal, as therapy animals, companions, as well as guardians of livestock. Their important contribution to science is yet another reason to appreciate our camelid friends.